|
6/16/2023 0 Comments Aluminum oxide reactivity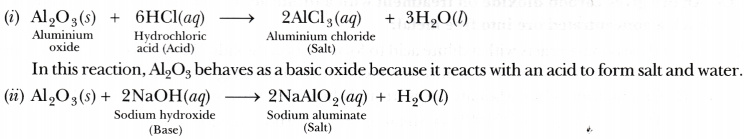
Aluminum oxide flakes are used in paints to add a reflective or ornamental appearance.Aluminum oxide makes around 5-10% of its makeup. Aluminum oxide is a frequent component of aluminosilicate glass. It is used in the manufacture of glass.It can be present in birth control pills and hip replacement surgery.It possesses excellent corrosion resistance and temperature stability, low thermal expansion, and a good stiffness-to-weight ratio. Due to its high electrical resistance, aluminum oxide is often used as the dielectric in capacitors to keep charges separated. Mechanical PropertiesĪluminum oxide has a high wear resistance since it is an extremely hard material, nearly as hard as diamonds. Some examples of these applications may include metering devices, wire guides, machinery gaskets, and high-temperature electrical insulators. Industrial PropertiesĪluminum oxide may be manufactured or cast into robust, wear-resistant composites for various engineering applications. Because of this, aluminum is referred to be a corrosion-resistant metal. This coating of aluminum oxide assists in the prevention of aluminum corrosion. Aluminum reacts with oxygen in the environment to form an aluminum oxide protective layer on its surface. Despite being particularly reactive to chlorine trifluoride and ethylene oxide, most compounds do not react quickly with aluminum oxide.Ī fire occurs when aluminum oxide is combined with one of these substances. The degree of resistance varies with the purity of the substance. In contrast to its relative aluminum, Al 2O 3 exhibits considerable electrical resistance. While being an electrical insulator in ceramics, aluminum oxide possesses heat conductivity.Īlso Check: Aluminium Chemical Properties.Aluminum oxide has a relatively high melting point (2345K) and boiling point (3250K).Aluminum oxide appears as a white powder.It is insoluble in water and nearly all other solvents.Aluminum oxide has a density of 3.987g/cm 3. 
Aluminum oxide is a white powdered material with no discernible odor. Hence masks are suggested for extended exposure. Although it is non-toxic, airborne aluminum oxide dust can pose industrial dangers. General PropertiesĪluminum oxide is extraordinarily heavy a 1-meter by 1-meter cube of aluminum oxide weighs approximately 7,200 lbs. The oxygen ions present in the compound are closely packed in hexagonal form with the aluminum ions.Īluminum Oxide (Al 2O 3) has the following Lewis structure:.The trivalent ions of the aluminum present in the compound are octahedral.Corundum is generally thermodynamically stable.Aluminum oxide has a molar mass of roughly 101.96 g/mol.Īluminum oxide is used in various lights, including sodium-vapour lamps, and as an electrical conductor in small circuits in the burgeoning nanotechnology sector. Due to its hardness and high melting point, Al 2O 3 is used to produce aluminum metal as an abrasive and refractory material. This aluminum oxide is found in many precious gemstones like ruby and sapphire. Corundum, the naturally occurring form of aluminum oxide, is generally in the crystalline polymorphic phase – Al 2O 3. The most common form of aluminum oxide is aluminum (III) oxide. When aluminum and oxygen combine, they form a compound called aluminum oxide.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
